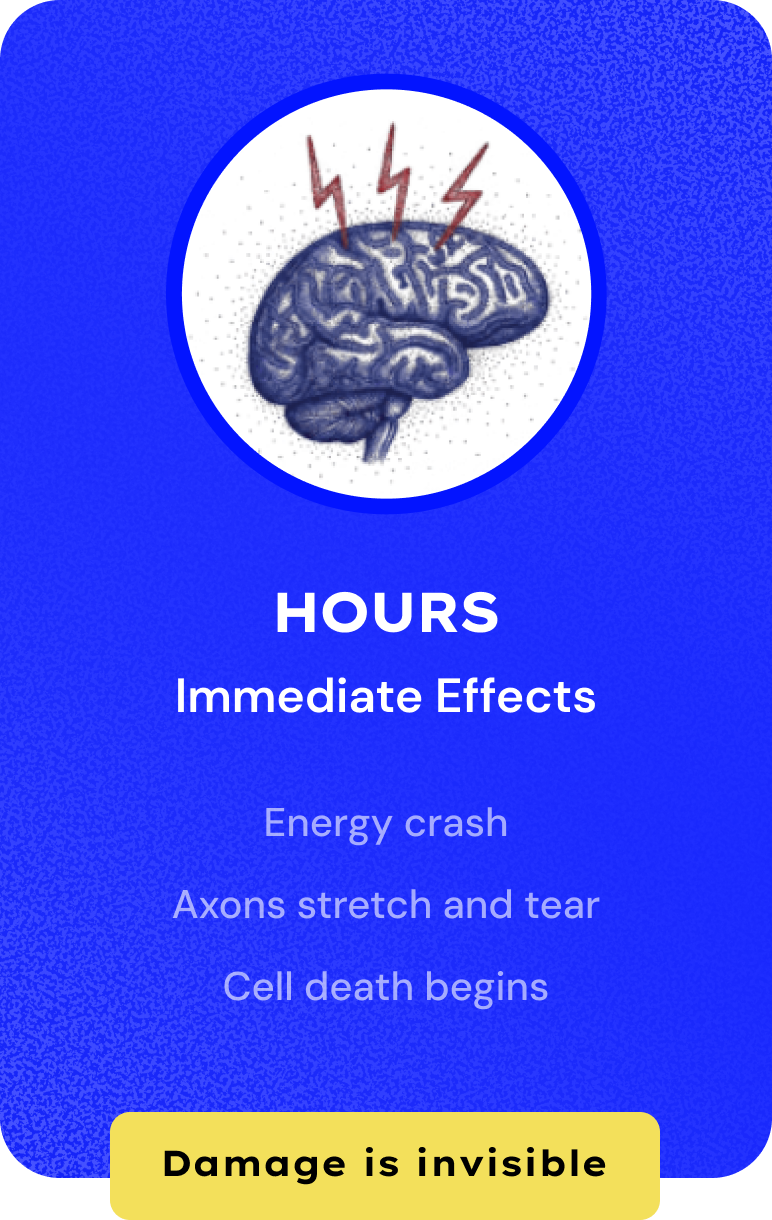
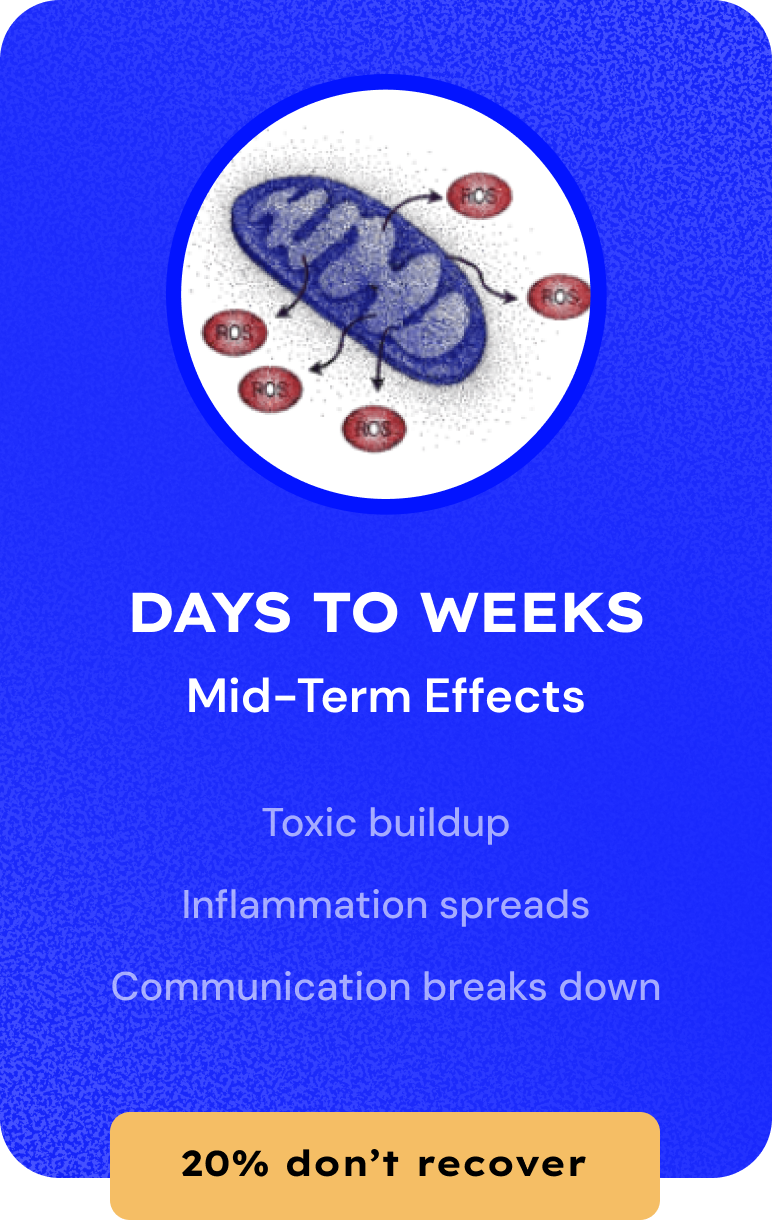
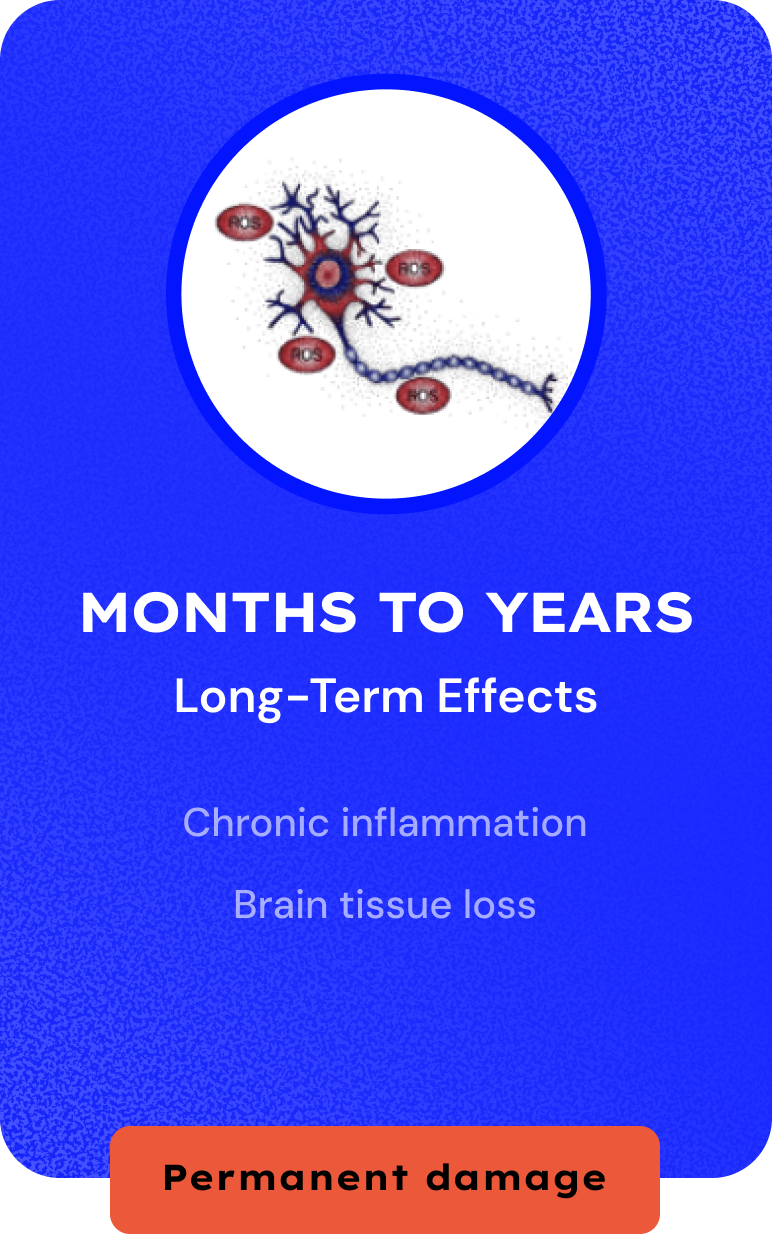
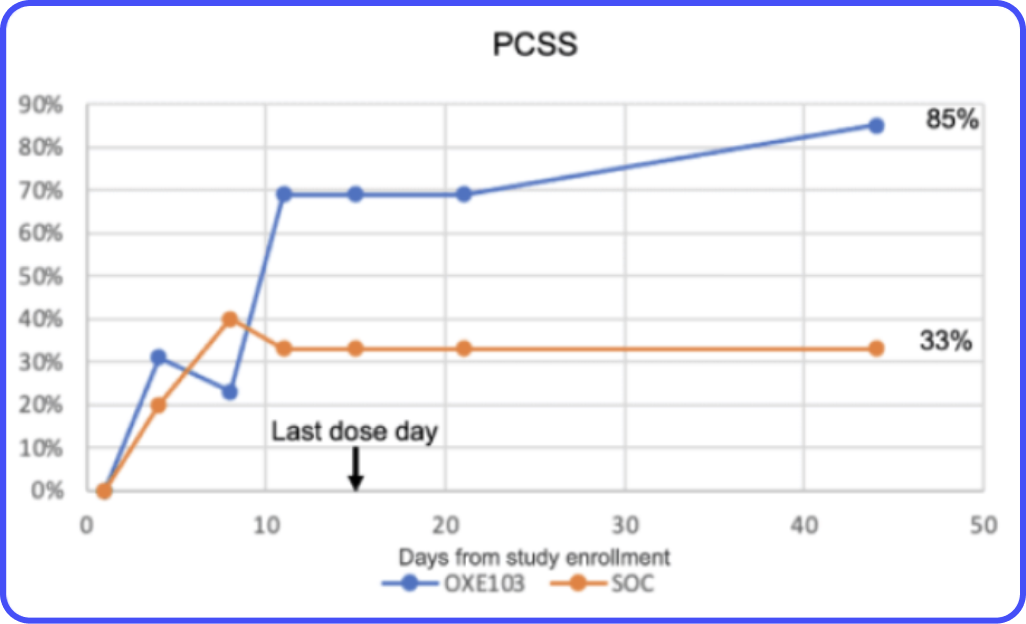
Millions Suffer from Concussion. Zero Treatments.
This Reg CF offering is made available through StartEngine Primary, LLC, a member of FINRA/SIPC. This investment is speculative, illiquid, and involves a high degree of risk, including the possible loss of your entire investment.
OXE103 is an investigational therapy subject to regulatory review and clinical development risk. Statements regarding regulatory approval, clinical development timelines, market opportunity, and potential outcomes are forward-looking and involve risks and uncertainties. Actual results may differ materially.